Maximize Output on Your Line with PerfectPeel™

In high-speed filling and sealing operations, maintaining output, efficiency, and consistency isn’t always straightforward. When lidding film requires higher temperatures and longer dwell times, it can slow production, increase energy use, and introduce process variability.
Scaling a Nutraceutical Brand Through Smarter Packaging

For Progressive Health, packaging plays a critical role in how their products perform. With a growing portfolio of targeted supplements and frequent product updates, they needed a packaging partner that could keep up with evolving SKUs, formats, and fast turnaround demands.
AWT Labels & Packaging Showcases Sustainable Innovation

AWT Labels & Packaging will be demonstrating our commitment to sustainability by handing out personalized ForestNation Tree Seed Packets again at the 2026 Natural Products Expo West Show, Booth #2911 in Anaheim, California.
Choosing the Right Clinical Trials Labeling Partner: A Sponsor’s Guide

When it comes to clinical trials, the importance of precision and accuracy cannot be overstated. Every detail counts, and this extends into what may seem like the minutiae of trial management: the labeling of clinical trial materials.
Maximize Line Efficiency with AWT PerfectPeel™
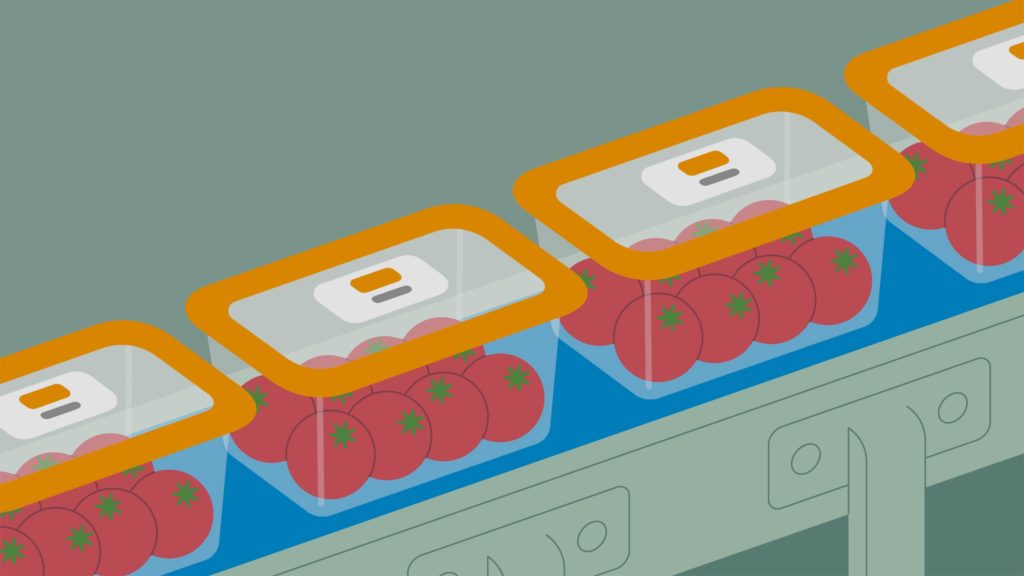
If your sealing station is slowing things down, you’re not alone. High temperatures and long dwell times can create bottlenecks, waste energy, and add unnecessary wear to your equipment.
PerfectPeel™ Lidding Film Helps You “Lose Weight” & Reduce Shipping Cost

Not only is PerfectPeel™ One a sustainable option from a plastic reduction/ weight loss standpoint, but there’s even more!
From Shrink Sleeve to Labels

From Shrink Sleeves to Labels. Whether you’re managing hundreds of SKUs, responding to new regulations, or elevating your brand’s shelf presence, packaging flexibility has never been more critical.
Major Changes Coming to How2Recycle Labels: Keeping You Up-to-Date

Using more traditional suppliers and methods, La Prenda would only order labels a handful of times per year in an effort to meet minimums. This was complicated by the dynamic needs of their customers, who were often looking for small lots and unique wines.
Is Your Color Always On Target?

AWT helps fine-tune your print registration so your labels always hit the mark. Whether you’re in food, beverage, household products, or personal care & beauty, we’re here to keep your colors naturally on target.

